According to FDA guidelines (Guidance for Industry, Analytical Procedures and Methods Validation, FDA, 2000), a Stability Indicating Method (SIM) is defined as a validated analytical peptide testing procedure that accurately and precisely measure active ingredients (drug substance or drug product) free from process impurities, excipients and degradation products. The FDA recommends that all assay procedures for stability should be stability indicating. The main objective of a stability indicating method is to monitor results during stability studies in order to guarantee safety, efficacy and quality. It represents also a powerful tool when investigating out-of-trend (OOT)) or out-of-specification (OOS) results in quality control processes.
Step 1: Generation of degraded samples for testing selectivity of the method
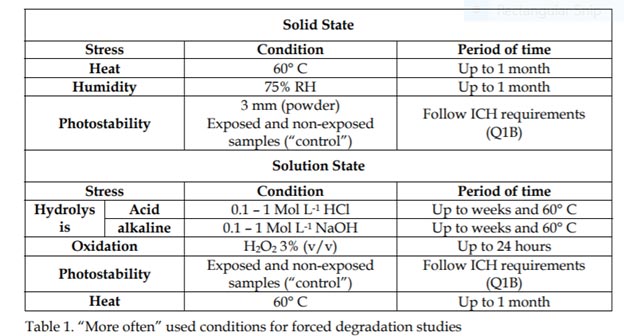
Stress tests should generate representative samples to assess drug substance and drug product stability, provide information about possible degradation pathway and demonstrate the stability indicating the power of the analytical procedures applied. Determination of Limit of Quantification (LoQ) in close relation to the determination of the amount of degradation is the evaluation of Limit of Detection (LoD) and Limit of Quantification (LoQ) of the method. These limits should be closely related to the Reporting, Identification
Step 2: method development (manipulating and evaluating selectivity/specificity)
Liquid chromatography is the most appropriate technique for developing/validating a SIM. The use of a diode-array-detector and, additionally, mass spectrometers, gives the best performance for people working with SIM development. The goal is to manipulate selectivity by changing mobile phase composition, the wavelength of detection and pH. Related to mobile phase pH, it can be said that the advances in LC column technology have made possible the use of pH as a true selectivity tool for the separation of ionizable compounds. Columns that are mechanically strong, with high efficiency and that operate over an extended pH range, should be preferred. Acidic compounds are retained more at low pH; while basic compounds are retained more at higher pH (neutral compounds are unaffected). At traditionally used pH values (pH 4 – 8), a slight change in pH would result in a significant shift in retention. Type of chromatography used (e. g. HPLC or GC) and arrangements/detectors (GC/FID, GC/MS, LC/DAD or LC/MS) are certainly useful tools. For HPLC, different modes of chromatography can be used (normal or reversed phase, ion pair or HILIC). Another powerful tool is the use of Light Scattering Detector (LSD) coupled to HPLC to monitor compounds without light absorption in the uv/Vis region. Gas chromatography may only be used when no additional thermal degradation of the sample is produced (sample inlet works on high temperatures). The use of HLPC coupled to diode-array detectors (DAD) in the achievement of peak purity usually gives reasonable results, mainly related to a reliable determination of the main active ingredient. It is possible to guarantee no co-elution with degradation peaks and other impurities. Indeed, the main feature of DAD detectors is that it is possible to collect spectra across a range of wavelengths at each data point collected across a peak, and through software manipulations involving multidimensional vector algebra, to compare each of the spectra to determine peak purity. In this manner, DAD detectors can distinguish spectral and chromatographic differences not readily observable by simple overlay comparisons. DAD detectors can be limited on occasion the more similar the spectra, and the lower the relative absorbance, the more difficult it can be to distinguish co-eluted compounds. MS detection overcomes many of these limitations. MS can provide unequivocal peak purity information, exact mass, structural and quantitative information depending upon the type of instrument used. MS is also a very useful tool to track peaks to selectivity manipulations in method development. As a disadvantage, MS detectors cannot handle non-volatile buffers, which are frequently used during the mobile phase in drug analysis. The combination of both DAD and MS on a single instrument and software platform provides the type of valuable orthogonal information required when evaluating specificity on SIM development. After determination of peak purity, in fact, the identification of degradation products and also mass balance determination usually are more complexes steps of analytical development, as in most cases, commercial reference standards of degradation products are not available.
Step 3: Method validation
Validation is not an efficient way to do method development so efforts should be dedicated in the specificity step of the method, working with the stressed samples. The validation routine may start with a protocol based on pharmacopeia and/or ICH guidelines (Q2B). For assay procedures, that are intended to measure the analyte present in a given sample, typical validation items should be considered: Accuracy, Precision (repeatability and intermediate precision), Specificity, Detection and Quantitation Limits, Robustness, Linearity and Range.
References
- “Draft Guidance for Industry on Analytical Procedures and Methods Validation: Chemistry, Manufacturing, and Controls Documentation.” Federal Register. 30 Aug. 2000. Retrieved on 2 Dec 2021, https://www.federalregister.gov/d/00-22143.
- Cione, Ana Paola et al. “Stability Indicating Methods.” (2011).
Interested in learning more about AmbioPharm Peptide Production and Services? Contact Us or read more about us and the API model.

